Website cases:
CASE NUMBER 31
[DigitalScope]
Clinical History: A 64-year-old man with history of uncontrolled diabetes and hypertension presented to the emergency department with a 10-hour history of severe chest pain that radiated to both arms and his neck. It was accompanied by diaphoresis and dyspnea.
On exam: BP 160/100, HR 110, RR 24, O2 98% on room air. Heart rate was regular and there was trace bilateral lower extremity edema. A high sensitivity troponin assay showed a level of 1000 ng/L (normal, <14 ng/L) and an ECG showed 3 mm of ST elevation in leads V2-V4.
The patient was taken to the cardiac catheterization lab for percutaneous intervention with a diagnosis of ST elevation myocardial infarction (STEMI). Afterwards, he was admitted to the hospital. Three days after admission, he experienced an episode of ventricular fibrillation and died. An autopsy was performed.
Image Gallery:
|
Review Heart Histology
Slide 13: Heart
[DigitalScope]
Normal heart tissue sections demonstrate no evidence of fibrosis or hemorrhage. Cardiac myocytes have moderately sized centrally located nuclei. Normal myocytes are not brightly eosinphilic. Normally no inflammation is seen. Normal cardiac myocytes do not show hypertrophy.
|
Summary of Gross Findings
The heart was slightly enlarged weighing 460gms. There was severe atherosclerosis of all the major coronary arteries with a recent thrombotic occlusion of the proximal left anterior descending coronary artery. A recent transmural infarct was present in the left ventricle that involved the interventricular septum and the papillary muscle.
|
Summary of Microscopic Findings
The slide includes a transmural section of the left ventricle. Nearly the entire section is involved by infarct. However, there is a thin rim (5 to 10 cell layers) of endocardial myocytes which have survived because of diffusion of oxygen and nutrients from the ventricular cavity. Other viable myocytes can be found around larger blood vessels within the section. The intense hypereosinophilia of the necrotic myocytes can best be appreciated by comparing the thin rim of lighter staining subendocardial myocytes with the deeper cells. Note also the karyolysis that is characteristic of coagulation necrosis. In some areas there is little inflammatory response. This observation is explained by microvascular necrosis which does not allow access of circulating leukocytes to these areas. In other areas, especially in the epicardial half of the infarct, there is an intense acute inflammatory response. Many intact neutrophils can be seen. In addition, there are many nuclear fragments from lysed neutrophils. Macrophage activity is not evident. These features of the inflammatory response indicate that the infarct was approximately three to four days old. Note also that the inflammation extends to the epicardial surface and that there are deposits of fibrin on the epicardium. This is called fibrinous pericarditis. The granular grey material seen within some blood vessels is barium sulfate, which was injected to permit post-mortem study of the coronaries by radiography.
|
Gross Image Questions:
- Identify (i.e., in an annotated image): right ventricle, left ventricle, interventricular septum, epicardial fat, normal myocardium, and area of infarct
Virtual Microscope Slide Questions:
- Identify (i.e., in an annotated image): viable myocytes, necrotic myocytes, nuclear pyknosis, a blood vessel, and epicardial fat.
- Why is there so much space between bundles of cardiac myocytes?
--ANSWER--
- Why do many of the neutrophils appear fragmented?
--ANSWER--
Differential Diagnosis:
- What is the differential diagnosis based on the initial presentation?
--ANSWER--
- What is the differential diagnosis following physical exam and laboratory testing?
--ANSWER--
31-1. Which of the following best describes the pathologic findings?
- Caseous necrosis
- Coagulative necrosis
- Fat necrosis
- Fibrinoid necrosis
- Gangrenous necrosis
--ANSWER--
31-2. Which of the following best describes the nuclear changes seen in the necrotic cardiac myocytes?
- Chromatin condensation
- DNA cleavage into nucleosomal subunits
- Karyorrhexis
- Karyolysis
- Pyknosis
--ANSWER--
31-3. Increased eosinophilia of the dead myocytes is most directly linked to which of the following molecular events?
- Cytoplasmic blebbling
- Denaturation of cytoplasmic proteins
- Increased cytoplasmic RNA
- Generation of myelin figures
- Loss of glycogen particles
--ANSWER--
31-4. Which of the following processes did the necrotic cells in this specimen undergo?
- Caspase activation
- Cell shrinkage
- Chromatin condensation
- DNA cleavage into nucleosomal subunits
- Loss of membrane integrity
--ANSWER--
31-5. Which of the following processes may have extended the area of cardiac necrosis following initiation of thrombolytic therapy?
- Direct toxic effect of the thrombolytic therapy
- Reperfusion injury
- Second thrombus in the same site
- Systemic hypotension
- Ventricular rupture
--ANSWER--
31-6. When the patient’s artery was initially occluded by thrombus, ischemia led to a rapid decrease in intracellular ATP levels. Which of the following is a consequence of this process?
- Efflux of calcium
- Detachment of ribosomes
- Increased pH
- Influx of potassium
- Decrease in anaerobic glycolysis
--ANSWER--
31-7. In this ischemic environment, the mitochondria are unable to maintain normal oxidative phosphorylation, resulting in release of reactive oxygen species (ROS) into the cytoplasm. Which of the following is the most direct consequence of this release?
- Apoptosis
- Failure of the Ca2+ pump
- Increased phospholipid breakdown
- Leakage of metabolites into the extracellular space
- Lipid peroxidation of membranes
--ANSWER--
CASE NUMBER 85 (slide 31557) - slide courtesy of UMich:
[DigitalScope]
Clinical History: A 64-year-old woman with a BMI of 43 was admitted to the emergency department with a 6-hour history of nausea, vomiting and jaw pain. An electrocardiogram showed ST segment elevation. A high sensitivity troponin assay showed a level of 1115 ng/L (normal <14 ng/L) and, following evaluation, the patient was treated with stenting of two coronary arteries. Additional laboratory values were significant for:
- alanine aminotransferase (ALT) of 36 U/L (normal 8-20 U/L)
- aspartate aminotransferase (AST) of 32 U/L (normal 8-20 U/L)
- lactate dehydrogenase (LDH) of 208 U/L (normal 45-90 U/L)
- alkaline phosphatase of 135 U/L (normal 20-70 U/L).
In view of her abnormal liver function tests, an abdominal CT was performed at the time of hospitalization. The liver was noted to be grossly enlarged without mass lesions. A gross image of the liver from a patient with a similar disease is provided (liver weight 1980 grams). A biopsy was performed.
Image Gallery:
|
Review Liver Histology
Slide 3: Liver
[ImageScope] [DigitalScope]
The liver is the organ that metabolizes nutrients received from the digestive tract. These nutrients and processed by tissue hepatocytes which are large polygonal cells. The hepatocyes are separated by portal triads. The triads consist of an artery, a vein and a bile duct. The bile duct is lined by cuboidal epithelium. The artery has a muscular wall and a flat endothelial lining. The sinuses are well defined and contain a small amount of blood.
|
Summary of Gross Findings
The liver weighed 1980 grams. It had a pale, greasy appearance.
|
Summary of Microscopic Findings
The areas of fatty change are readily visible with use of scanning power. They are zonal. Note the peri-portal areas are severely involved and the areas of fatty change extend from one portal area to another. The more severely involved cells have eccentric nuclei and the entire cytoplasm is replaced by a fat globule.
|
Gross Image Questions:
- Describe the general appearance of the liver. Is there a focal lesion or is the process diffuse?
--ANSWER--
Virtual Miscroscope Slide Questions:
- Is there any normal liver present?
--ANSWER--
- Identify (i.e., in an annotated image) a portal triad and a central vein.
--ANSWER--
The central veins of the liver (or central venules) are veins found at the center of hepatic lobules (one vein at each lobule center). They receive the blood mixed in the liver sinusoids and return it to circulation via the hepatic veins.

Portal triads are collections of branches of: 1) the hepatic artery, 2) hepatic portal vein, and 3) bile duct found within "portal tracts" at the boundaries of hepatic lobules. In addition to the 3 elements above, lymphatic vessels and nerves are also found within the portal tracts

- Describe the pathologic findings. Identify (i.e., in an annotated image) the different zones of the acinus (aka the zones of Rappaport). Which zone is affected in this specimen?
--ANSWER--
The liver is showing focal areas of steatosis. Hepatocellular fat accumulation typically begins in centrilobular hepatocytes (zone 3). The lipid droplets range from small (microvesicular) to large (macrovesicular), the largest filling and expanding the cell and displacing the nucleus. As steatosis becomes more extensive, the lipid accumulation spreads outward from the central vein to hepatocytes in the midlobule and then the periportal regions (zone 1).

This microscopic appearance does not quite match the gross appearance, but we chose this one since you could appreciate the liver architecture and to bring back a little bit of HSF for you. In true non-alcoholic steatohepatitis, the liver is just stuffed full of fat and can be 4-6 kg. For a representative image, see [UMich 31102]
Differential Diagnosis:
85-1a. What is the differential diagnosis for this patient’s initial presentation?
--ANSWER--
85-1b. What is the differential diagnosis for her laboratory and radiologic findings?
--ANSWER--
85-2. Which other organs may be involved by this process?
--ANSWER--
85-3. Which of the following is the substance that has accumulated in the hepatocytes?
- Alpha-1-antitrypsin
- Carbon
- Cholesterol
- Lipofuscin
- Triglyceride
--ANSWER--
85-4. List several causes of this condition.
--ANSWER--
CASE NUMBER 3 (UMich slide 84)
[DigitalScope]
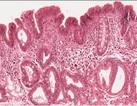
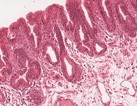
Clinical History: A 53-year-old man with a BMI of 44 presents to a free clinic. He reports that he has had “heartburn” for the last ten years that he has been treating with an over-the-counter proton pump inhibitor that has offered partial relief. He has no other symptoms. Endoscopy is performed with biopsy. Following biopsy analysis, an endoscopic mucosal resection is performed. However, surgical margins are found to be positive and a subsequent partial esophagogastrectomy is performed.
Image Gallery:
|
Review Esophagus Histology
Slide UCSF 226 (esophagus, H&E) [DigitalScope]
Slide UMich 126 40x (trachea & esophagus, H&E) [DigitalScope]
Slide UMich 153 20x (esophagus, H&E) [DigitalScope]
Slide UMich 155 40x (gastro-esophageal junct, H&E) [DigitalScope]
For the purpose of histological descriptions, the esophagus is subdivided into upper (entirely skeletal muscle in the muscularis externa), middle (mixed smooth and skeletal muscle) and lower (entirely smooth muscle) portions. Slide UCSF 226 is from the upper 1/3; slides 126 and 153 are from the middle 1/3; and slide 155 is from the lower 1/3 (at the esophageal-cardiac junction). The esophageal epithelium is the non-keratinized stratified squamous type and is supported by a connective tissue lamina propria. Note the presence of isolated lymphoid nodules and scattered leukocytes in the lamina propria. A rather thick layer of longitudinally arranged smooth muscle fibers form the muscularis mucosae. The connective tissue of the submucosa consists of mostly collagenous fibers with some elastic fibers and varying amounts of fat as well as submucosal sero-mucous glands which can be readily observed in both slide 126 and slide 153 (the glands in slide 155 are not very well preserved).
In the upper esophagus, as shown in Slide UCSF 226, the muscularis externa consists of both inner and outer layers of skeletal muscle only. In the middle esophagus, the muscularis externa contains a mixture of skeletal and smooth muscle as seen in slide 126, whereas in the lower esophagus only smooth muscle is found as seen in slide 155. Present in all regions of the esophagus (upper, mid, and lower) is the myenteric (Auerbach’s) plexus between the two layers of the muscularis externa. For most of its extent, the esophagus is retroperitoneal, so its outermost layer consists of a connective tissue adventitia which merges with the adjacent connective tissue associated with nearby structures (such as the trachea as shown in slide 126). Below the diaphragm, however, the esophagus is suspended within abdominal cavity and is therefore covered by a connective tissue serosa as shown in slide 155.
|
Summary of Gross Findings
Salmon-pink tongues of tissue are seen extending past the gastroesophageal junction, consistent with Barrett esophagus.
|
Summary of Microscopic Findings
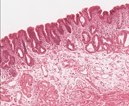
The slide is taken from the gastro-esophageal junction --the far left side of the slide shows slightly inflamed gastric mucosa and the remainder of the slide is of the esophageal mucosa (note the submucosal glands found under the thick muscularis mucosae that helps identify the esophageal portion of the specimen). Small pockets of stratified squamous epithelium can be seen as one would expect in the esophagus, but most of the esophageal mucosa consists of a columnar epithelium that has scattered deep mucus-producing glands and superficial elongated tubules, some of which extend from the surface to the base of the mucosa. Some of these tubules are lined by gastric-type surface epithelium with apical pink mucous vacuoles. Other tubules have intestinal-type goblet cells with big blobs of pale-staining mucus, evidence of intestinal metaplasia. Still other tubules have very little mucus of any type and contain epithelium with large, stratified nuclei which, in a few areas, are hyperchromatic and even vary in size and shape. These latter features are those of dysplasia.
|
|
Gross Image Questions:
- In the endoscopic image from the patient, identify (i.e. in an annotated image) the lesion. Identify normal tissue.
Virtual Micropscope Slide Questions
- Identify (i.e., in an annotated image) the lesion, normal tissue, gastric mucosa, and esophageal mucosa.
3-1. What are the significant endoscopic findings?
--ANSWER--
3-2. What are the significant histologic findings?
--ANSWER--
Differential Diagnosis:
3-3a. What is the differential diagnosis based on the patient’s initial presentation?
--ANSWER--
3-3b. What is the differential diagnosis based on the endoscopy findings?
--ANSWER--
3-3c. What is the differential diagnosis based on the histology findings?
--ANSWER--
3-4. Which of the following is the appropriate term for the pathologic process that accounts for the presence of goblet cells in this specimen?
- Anaplasia
- Dysplasia
- Hyperplasia
- Hypertrophy
- Metaplasia
--ANSWER--
3-5. Which of the following is the most likely cause of this process?
- Exposure to toxins
- Ischemia
- Low pH of stomach secretions
- Mechanical trauma
- Vitamin A deficiency
--ANSWER--
3-6. Which of the following is the mechanism by which this process is thought to occur?
- Activation of ubiquitin-proteasome pathway
- Autophagy
- Increased protein synthesis and organelle assembly
- Reprogramming of stem cells
- Unfolded protein response
--ANSWER--
Review Items
CELL INJURY
Key Vocabulary Terms (click here to search any additional terms on Stedman's Online Medical Dictionary)
Enable java script to view definitions.
CELL INJURY GOALS and LEARNING OBJECTIVES
Goal 1: Cellular Response to Injury
Apply knowledge of membrane physiology, metabolism, signal transduction and macromolecular synthesis to discuss cellular responses to injury at the cell, tissue and organism levels; how these responses affect morphologic appearance; and how they can be used for diagnostic, prognostic, and therapeutic purposes.
- Objective 1: Necrosis
Define necrosis, and compare and contrast the different forms of necrosis (e.g. coagulative, fibrinoid, caseous, liquefactive, gangrenous, fat necrosis) produced in response to different etiologic agents with respect to their variable clinical and morphologic features.
- Objective 2: Ischemia
Compare and contrast ischemia and hypoxia, and discuss the time course of the molecular events that occur in a cell in response to lack of oxygen, emphasizing the events that distinguish reversible from irreversible injury.
- Objective 3: Reperfusion Injury
Summarize the cell’s response to reperfusion injury emphasizing how reperfusion can exacerbate injury produced by ischemia.
Goal 2: Cell Death
Apply knowledge of biochemistry and cellular physiology to differentiate between pathogenic and physiologic mechanisms of cell death, the resulting morphologic appearance and the physiologic and clinical settings in which these mechanisms are activated.
- Objective 1: Apoptosis
Contrast the etiology, mechanisms, and morphologic changes of apoptosis with those of necrosis, and discuss circumstances in which dysregulation of apoptosis can produce disease.
Goal 3: Sub-lethal Injury
Apply knowledge of cellular physiology, metabolism, and macromolecular synthesis to discuss cellular and subcellular responses to sublethal injury or stress on cells; how these responses affect morphologic appearance at the cell and tissue level; and how they can affect organ function.
- Objective 1: Cellular response to Environmental Changes
Discuss, with examples, changes that occur in cellular organelles or cytoskeletal proteins in different cell types in response to environmental alterations.
- Objective 2: Intracellular Accumulations
Describe the mechanisms of intracellular accumulations, and the morphologic and clinical consequences of these accumulations.
- Objective 3: Pathologic Calcification
Compare and contrast the physiologic basis, morphologic appearance and sites and associated diseases of metastatic and dystrophic calcification.
Goal 4: Cellular Aging
Apply knowledge of cellular physiology to discuss the process by which cells age.
- Objective 1:
Describe the etiology of cellular aging and the processes that lead to senescence.
Click here to submit questions or comments about this site.
Duke University | Duke Medicine | School of Medicine | School of Nursing | Doctor of Physical Therapy
Copyright © 2004-2026 Duke University School of Medicine
Updated 1/30/26 - Velkey |